We use cookies to make your experience better. To comply with the new e-Privacy directive, we need to ask for your consent to set the cookies. Learn more.
Share this :
Print
GenScript Trastuzumab Pharmacokinetic ELISA Kit
GenScript Trastuzumab Pharmacokinetic ELISA Kit - GSCRPT (Additional S&H or Hazmat Fees May Apply)
List Price
$498.00
Your Price
$498.00
HOW MUCH YOU SAVE:
0.00 %


| NETA PART: | GSCRPT-L00970 |
| MFG.PART: | L00970 |
| UNSPSC: | 41116126 |
| Manufacturer: | GenScript USA Inc |
Overview
| Product Description | Trastuzumab, also known as Herceptin, is a human epidermal growth factor receptor 2 (HER2) inhibitor for the treatment of breast cancer. Trastuzumab is a recombinant humanized IgG1 kappa monoclonal antibody. It binds to the extracellular domain of the HER2 protein on the on the surface of HER2-positive tumour cells with high affinity. Trastuzumab can suppress the cells growth and proliferation by blocking the ability of the cancer cells to receive chemical signals. |
Product Introduction
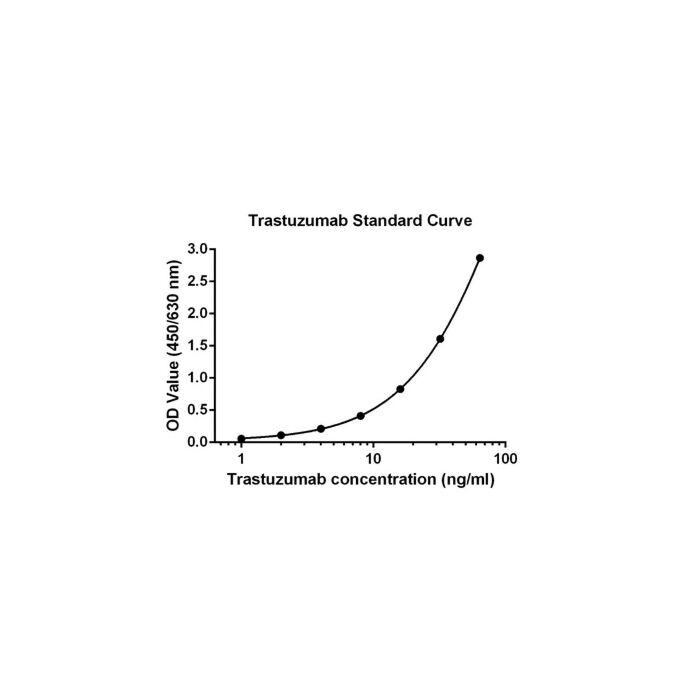
| LLOQ | 1 ng/mL | ||||||||||||||||||||||||||||||
| ULOQ | 64 ng/mL | ||||||||||||||||||||||||||||||
| Precision | Intra-assay: CV≤10% Inter-assay: CV≤15% | ||||||||||||||||||||||||||||||
| Minimum required dilution (MRD) | 1:40, validated non-human primate plasma | ||||||||||||||||||||||||||||||
| Kit Contents |
| ||||||||||||||||||||||||||||||
| Storage | The unopened kit is stable for at least 12 months from the date of manufacture at 2°C to 8°C, and the opened kit is stable for up to 1 month from the date of opening at 2°C to 8°C. |
Assay Principle
| Assay Principle | Trastuzumab Pharmacokinetic ELISA Kit is a sandwich ELISA assay with a pair of anti-idiotypic monoclonal capture and detection antibodies. When standards or samples are added to the capture plate, the Trastuzumab in the sample can be captured on the plate coated with the anti-Trastuzumab capture antibody. Then the Biotin Anti-Trastuzumab Antibody is added to interact with the Trastuzumab bound on the plate. Streptavidin-HRP (Streptavidin-Horseradish Peroxidase conjugate) is added to interact with the Biotin Anti-Trastuzumab Antibody. After the washing steps, TMB Solution is added, resulting in the formation of blue color. The reaction is stopped by adding Stop Solution. Adding the Stop Solution changes the color from blue to yellow. The intensity of the color can be read at 450 nm and 630 nm by a microplate reader. |
| Reference | 1. International Council for Harmonisation of TechnicalRequirements for Pharmaceuticals for Human Use. ICH Harmonised Guideline,Bioanalytical Method Validation M10 (2019). 2. US FDA. Bioanalytical Method Validation Guidance for Industry (2018). 3. European Medicines Agency. Guideline on Bioanalytical Method Validation(2011). 4. |
| SKU | GSCRPT-L00970 |
|---|---|
| Supplier Part Number | L00970 |
| UM | EA |
| UNSPSC | 41116126 |
| Manufacturer | GenScript USA Inc |
| ProductLine | GSCRPT |
| Qty | 1 |
| MinOrderQty | 1 |
| Weight | 7.00 |
| Lead Time | 7 Business Days |
| Hazardous | Y |